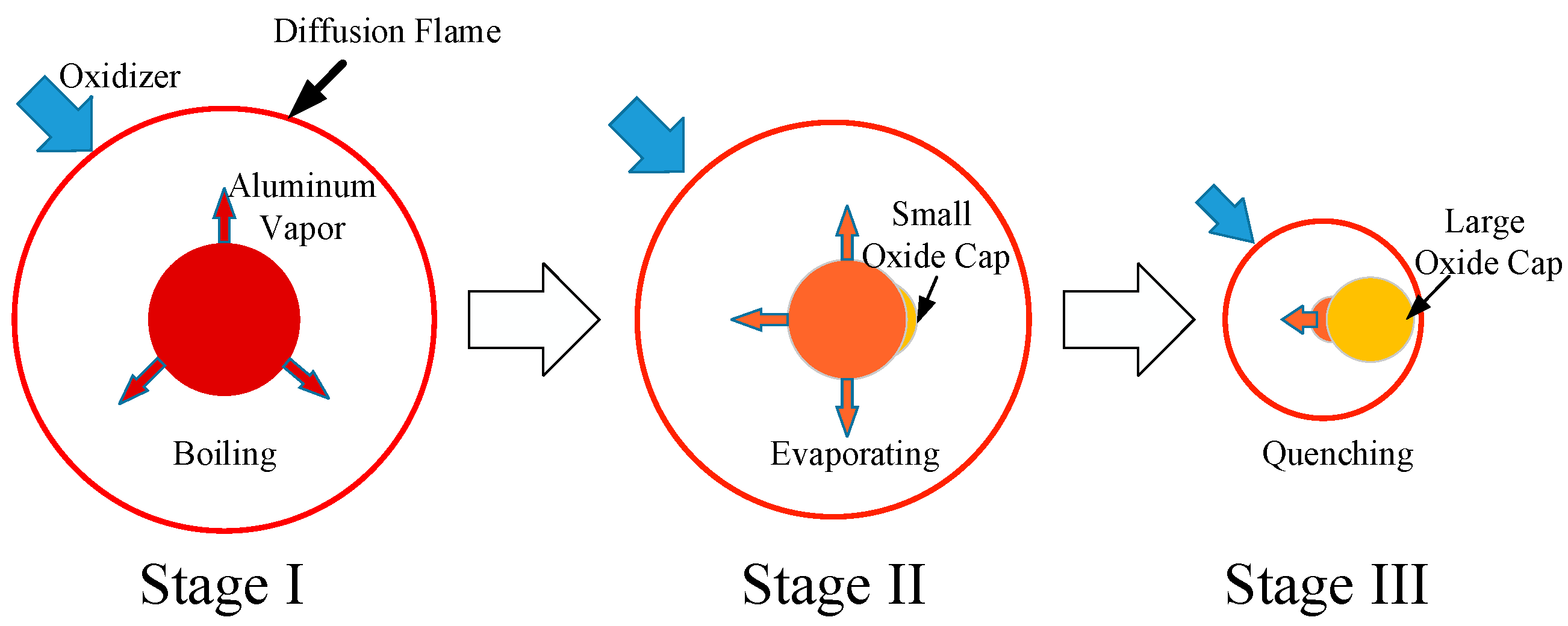
Applied Sciences | Free Full-Text | The Simulation of Different Combustion Stages of Micron-sized Aluminum Particles | HTML

On Modeling the Combustion of a Single Micron-Sized Aluminum Particle with the Effect of Oxide Cap | ACS Omega

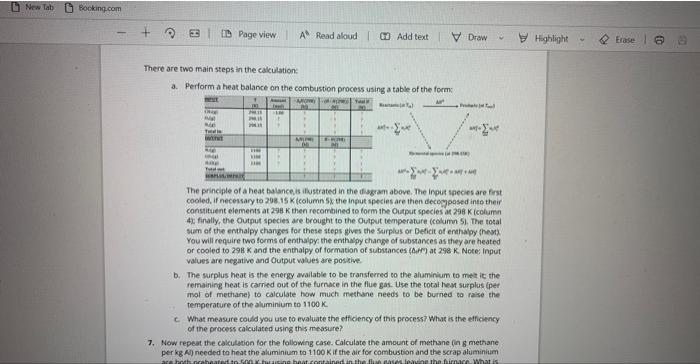
Consider the table below, showing the formation of the hydrogen halides, with their accompanying Δ H^∘ and Δ S^∘ values. Assume each reaction occurs at constant pressure.Reaction Δ H^∘ Δ S^∘ A

SOLVED:Write reactions for which the enthalpy change will be a. ΔHf^∘ for solid aluminum oxide. b. the standard enthalpy of combustion of liquid ethanol, C2 H5 OH(l) . c. the standard enthalpy
![PDF] Thermochemistry of aluminum species for combustion modeling from Ab Initio molecular orbital calculations | Semantic Scholar PDF] Thermochemistry of aluminum species for combustion modeling from Ab Initio molecular orbital calculations | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6b19c29b550295ebb7fa76e1ae5c20d8c3c951f5/5-Table1-1.png)
PDF] Thermochemistry of aluminum species for combustion modeling from Ab Initio molecular orbital calculations | Semantic Scholar

SciELO - Brasil - Theoretical and Experimental Heat of Combustion Analysis of Paraffin-Based Fuels as Preburn Characterization for Hybrid Rocket Theoretical and Experimental Heat of Combustion Analysis of Paraffin-Based Fuels as Preburn
![PDF] Thermochemistry of aluminum species for combustion modeling from Ab Initio molecular orbital calculations | Semantic Scholar PDF] Thermochemistry of aluminum species for combustion modeling from Ab Initio molecular orbital calculations | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6b19c29b550295ebb7fa76e1ae5c20d8c3c951f5/6-Table2-1.png)