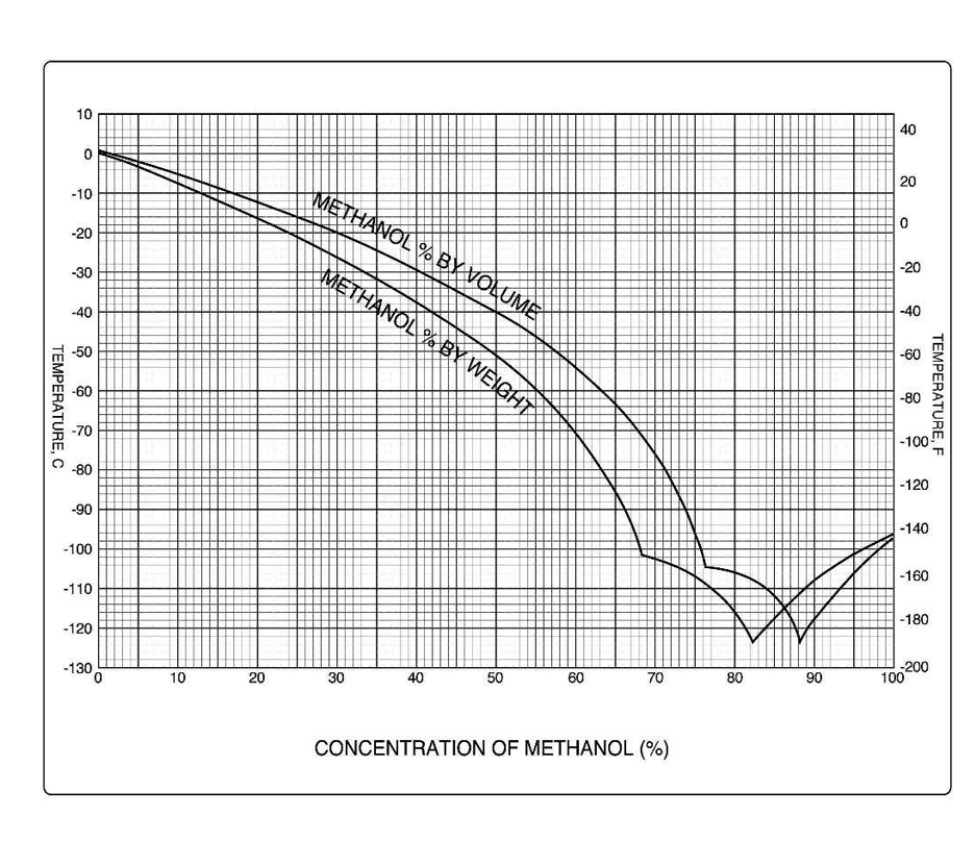
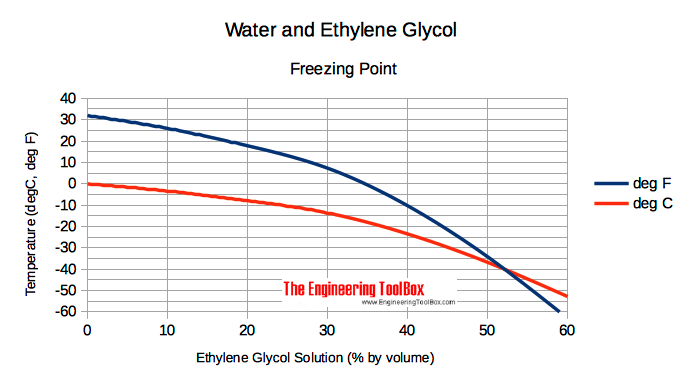
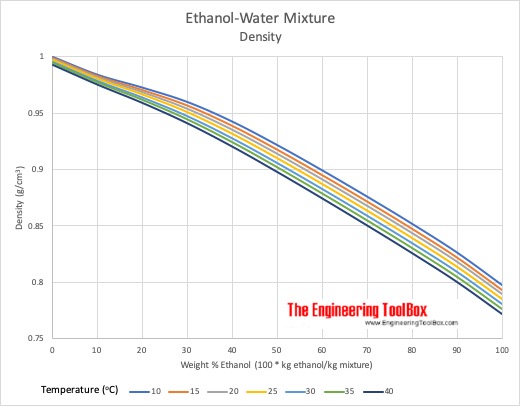
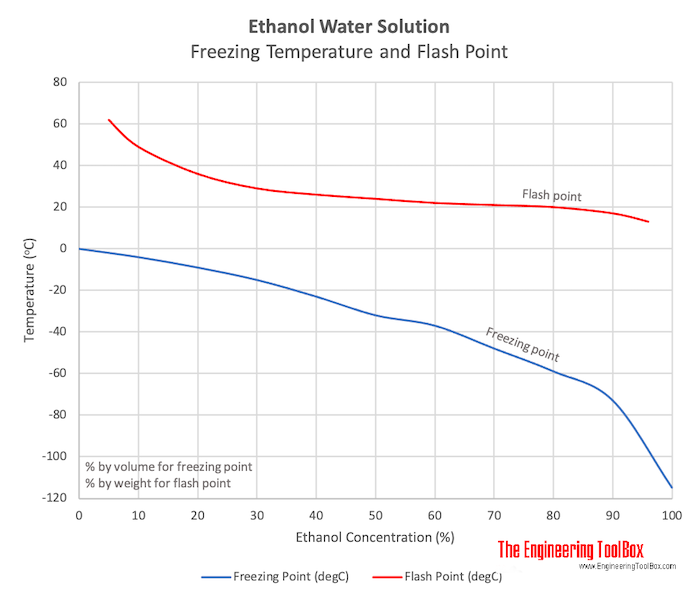
The relationship between the concentration of ethanol and its freezing... | Download Scientific Diagram
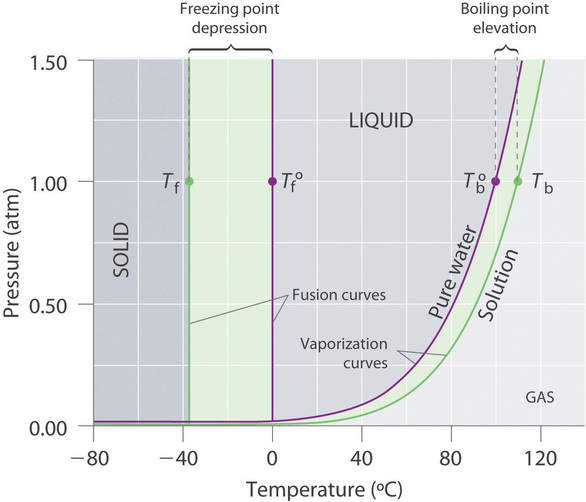
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts
I have a water-ethanol solution. What would happen if I keep it in the freezer? Will the water freeze and will I be left with pure ethanol? - Quora

Freezing Point Depressions of Aqueous MEA, MDEA, and MEA−MDEA Measured with a New Apparatus | Journal of Chemical & Engineering Data

thermodynamics - What mixing ratio of ethanol and acetone has the lowest freezing point? - Chemistry Stack Exchange
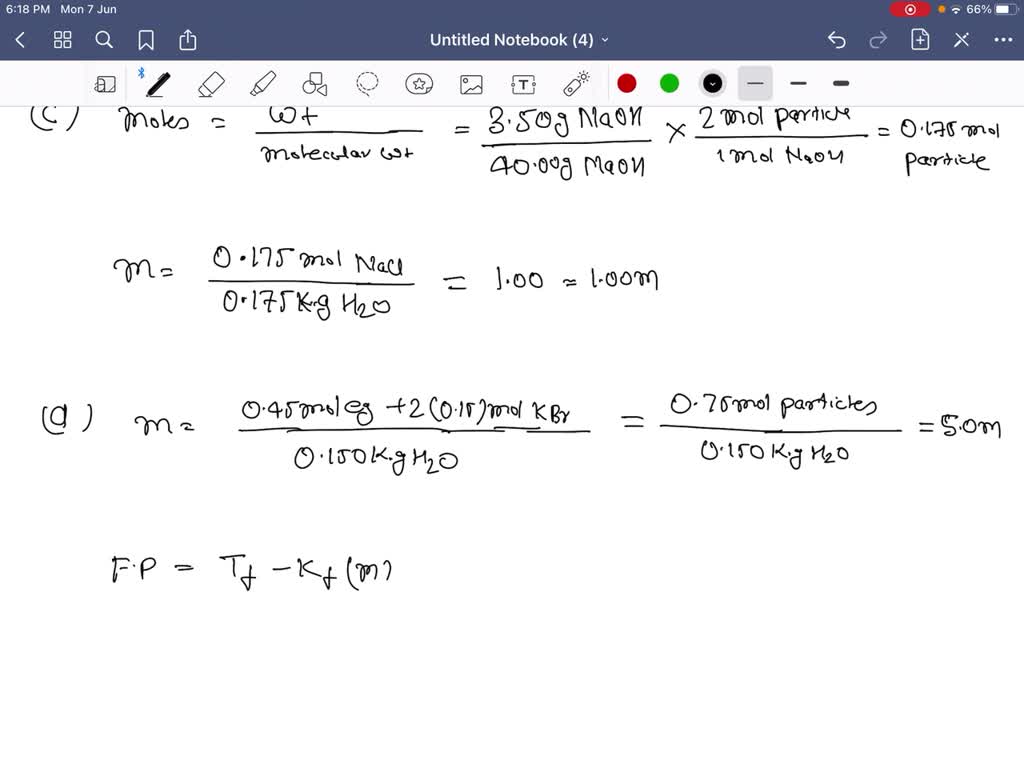
SOLVED:Using data from Table 13.3, calculate the freezing and boiling points of each of the following solutions: (a) 0.25 m glucose in ethanol; (b) 20.0 g of decane, C10 H22, in 50.0



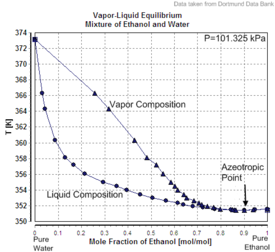
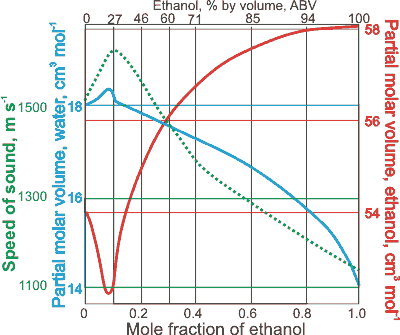
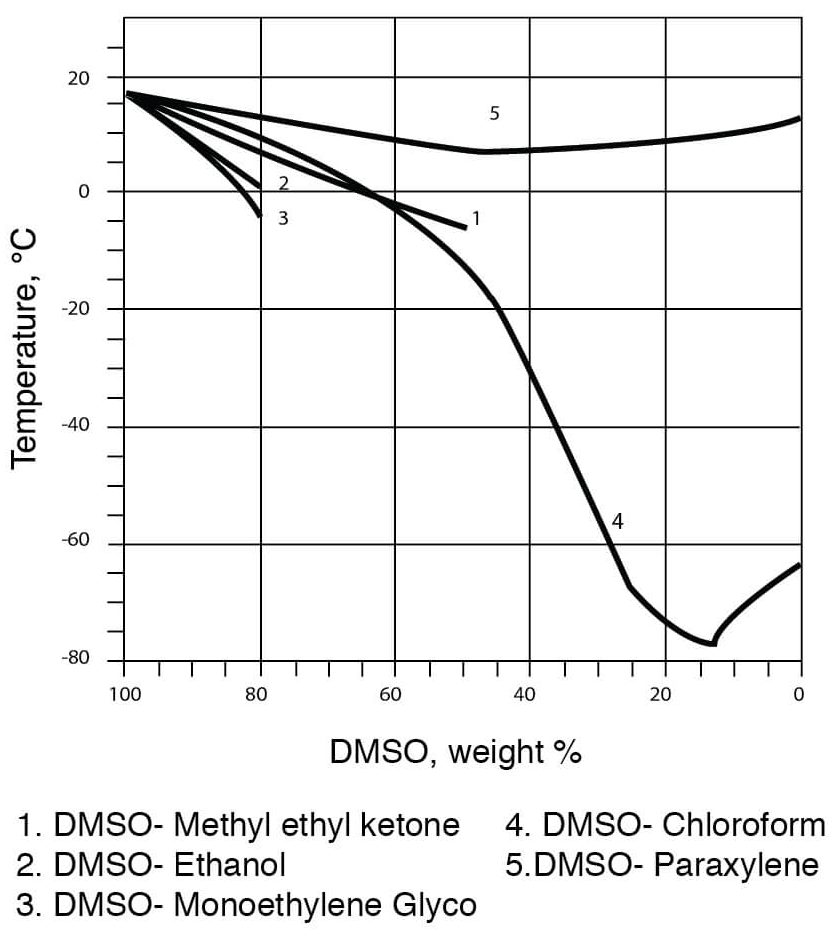
:max_bytes(150000):strip_icc()/will-liquor-freeze-760302_V3-1909ef0532f34ff0a2acee1a01b093c5.png)