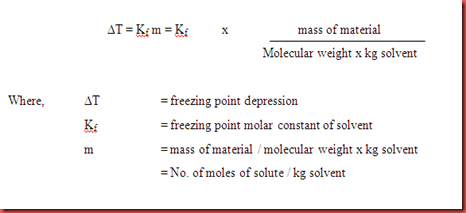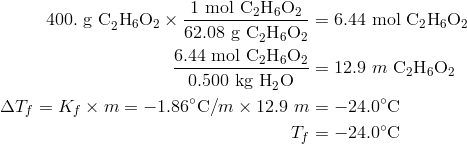A brief introduction to freezing point depression THE COLLIGATIVE PROPERTIES OF MOLALITY AND FREEZING POINT DEPRESSION grownextgen.org. - ppt download

Calculate the depression in freezing point of water when 10 g of CH(3)CH(3)CH(CI)COOH is added to 250 g of water. K(a)=1.4xx10^(-3), K(f)=1.86 K kg mol^(-1).

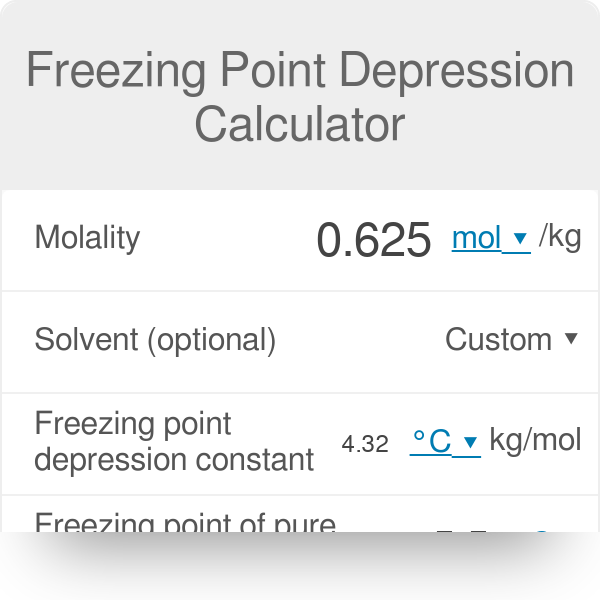
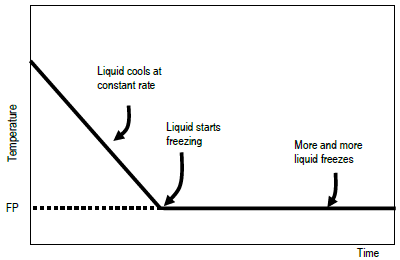
How do you find the freezing point of pure water from the freezing point depression equation? | Homework.Study.com

Calculations Involving Colligative Properties Freezing Point Depression and Boiling Point Elevation Calculations. - ppt download

Calculate the freezing point and the boiling point at 1 atmosphere of a solution containing 30 g cane sugar (molecular mass 342 ) and 150 g water.Given : Kb = 0.513 and Kf = 1.86

Calculate the freezing point depression and boiling point elevation of a solution of `10.0 g` of... - YouTube