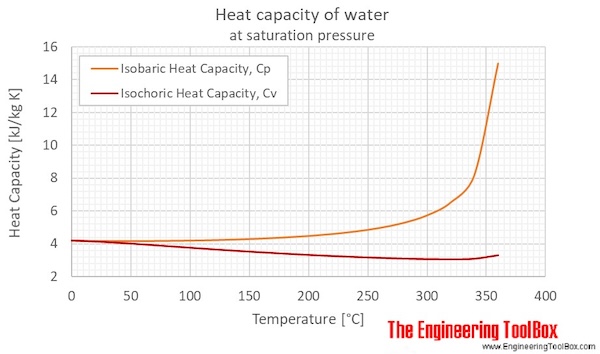
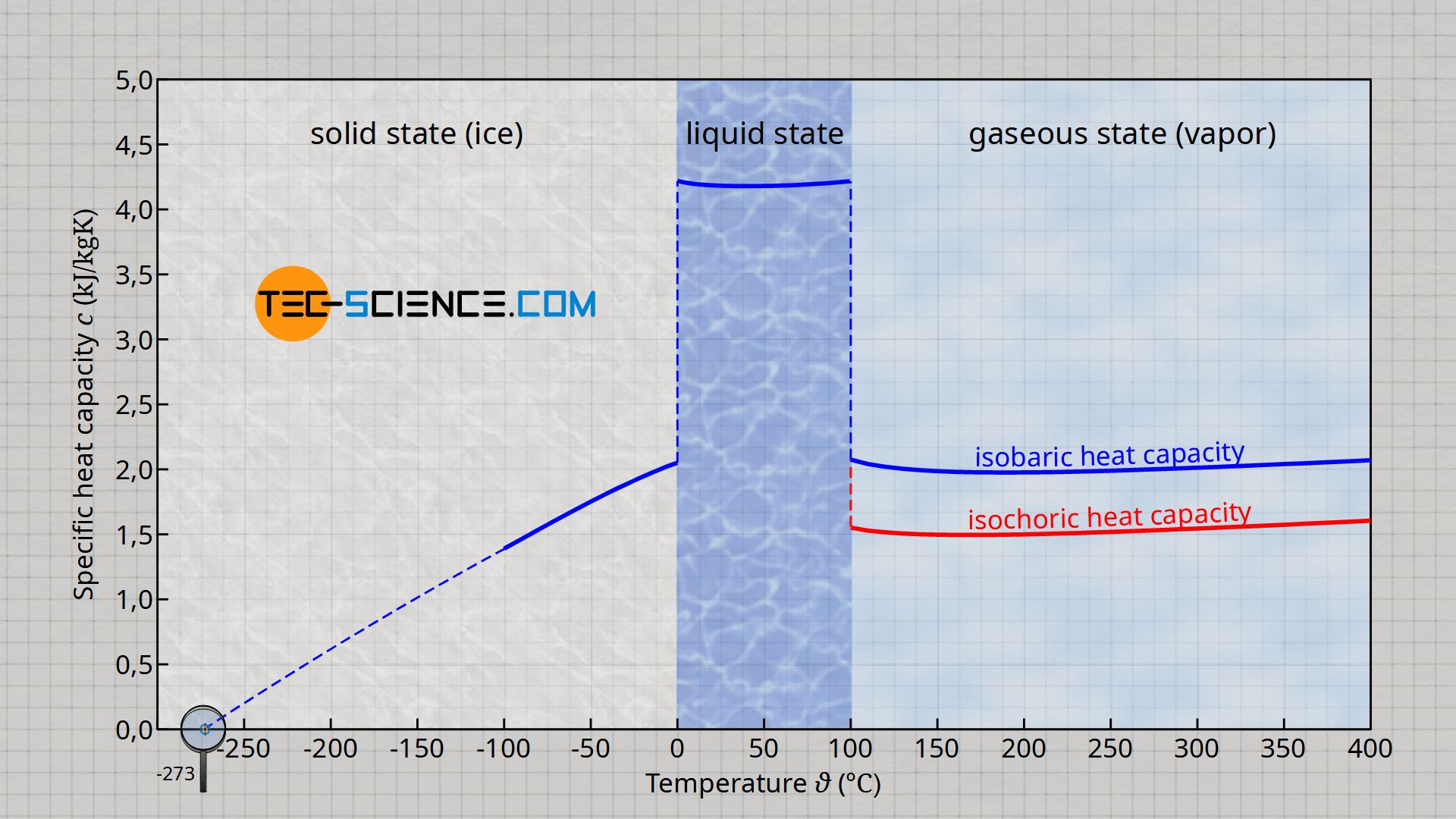
![PDF] Dependence of the isobaric specific heat capacity of water vapor on the pressure and temperature | Semantic Scholar PDF] Dependence of the isobaric specific heat capacity of water vapor on the pressure and temperature | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6c137407717ac2381ee1b2a89743eedede34ab16/4-Figure3-1.png)
PDF] Dependence of the isobaric specific heat capacity of water vapor on the pressure and temperature | Semantic Scholar
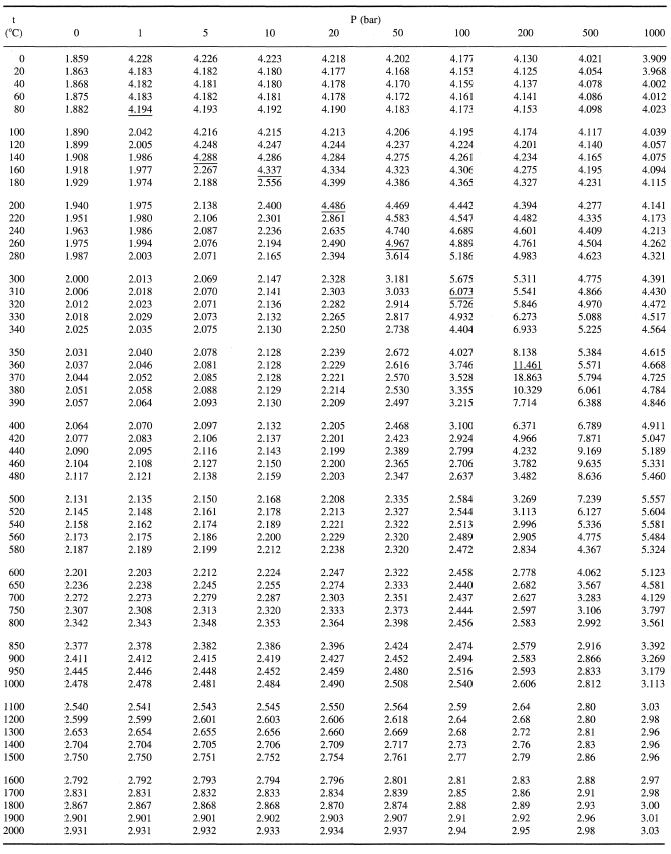
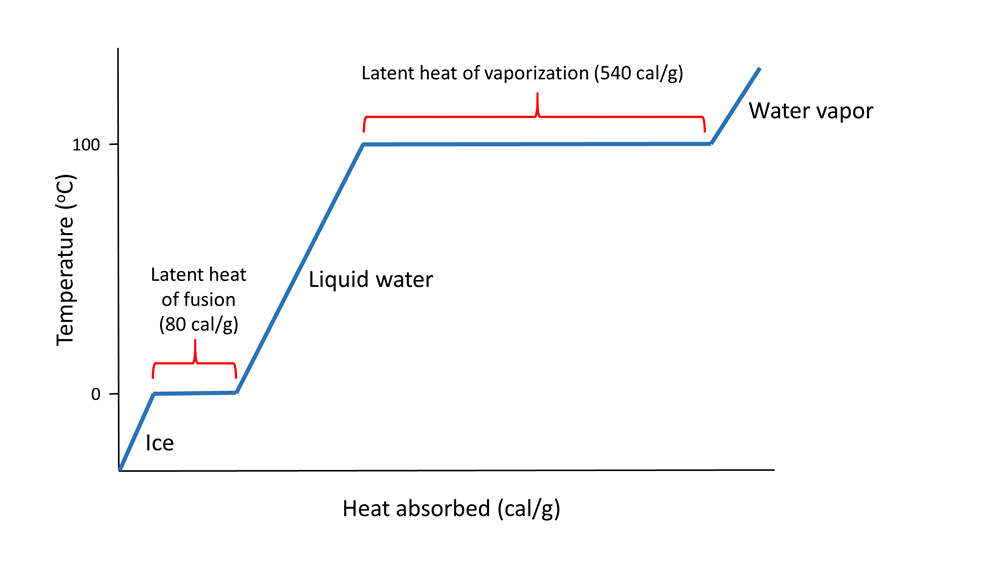
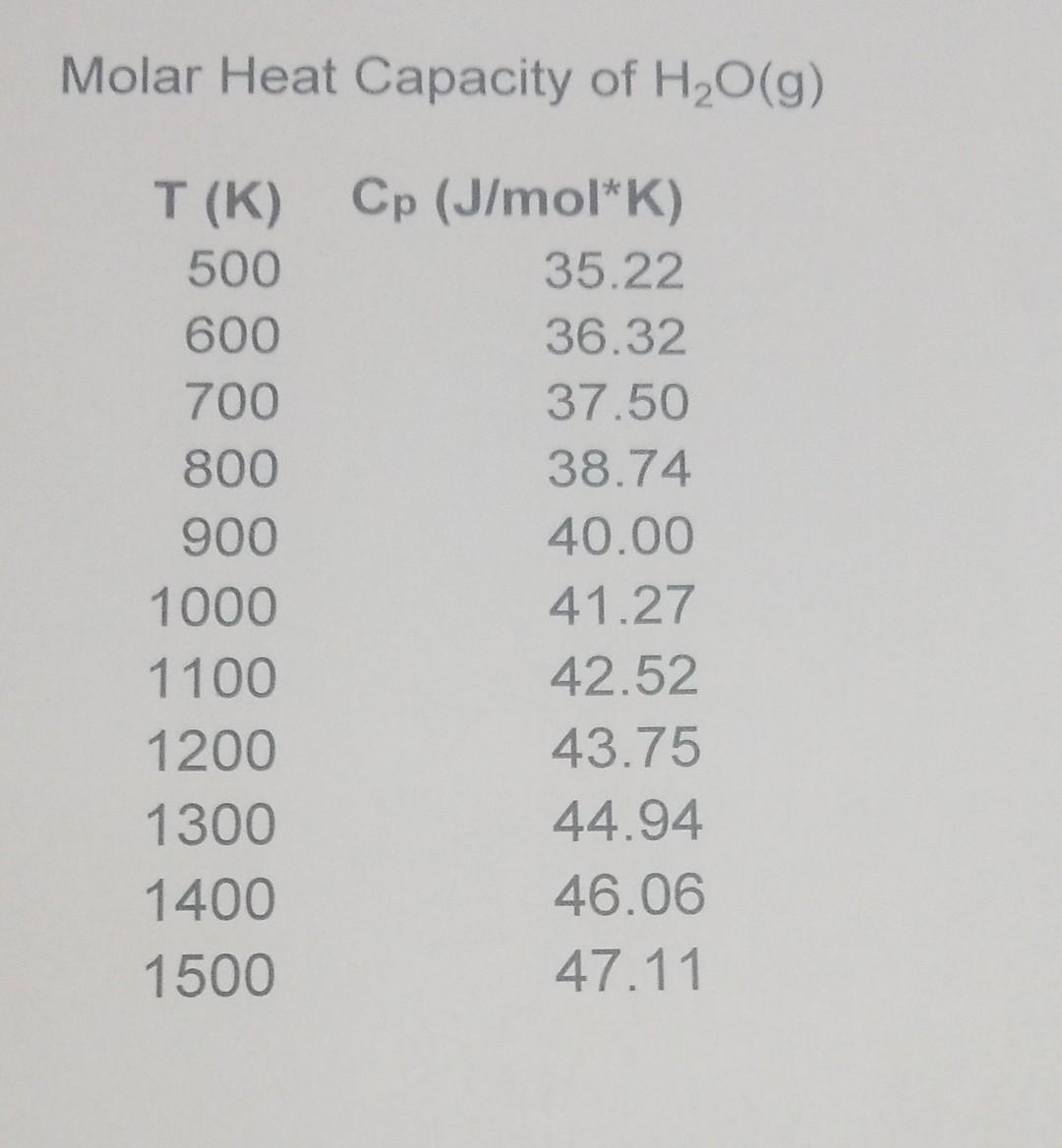
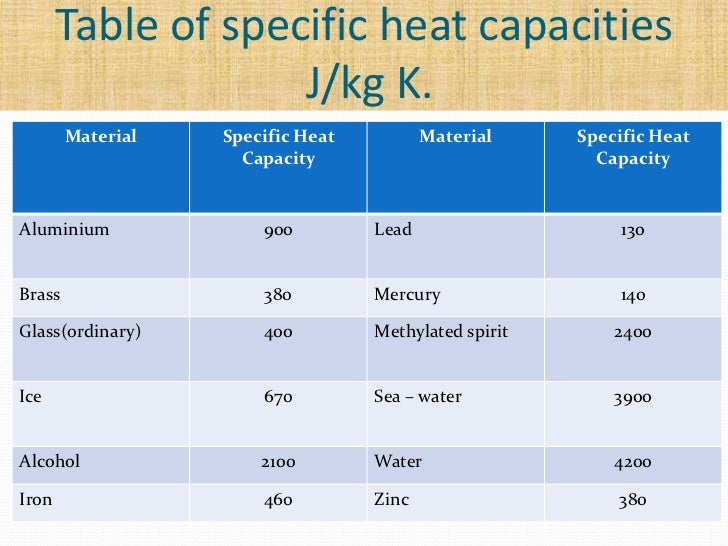
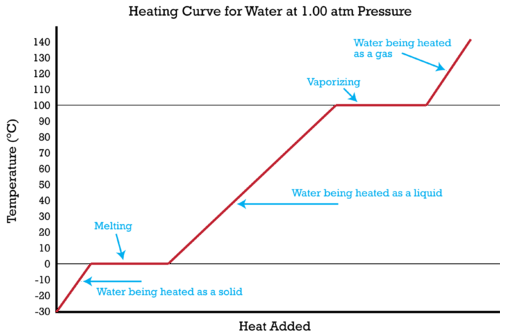
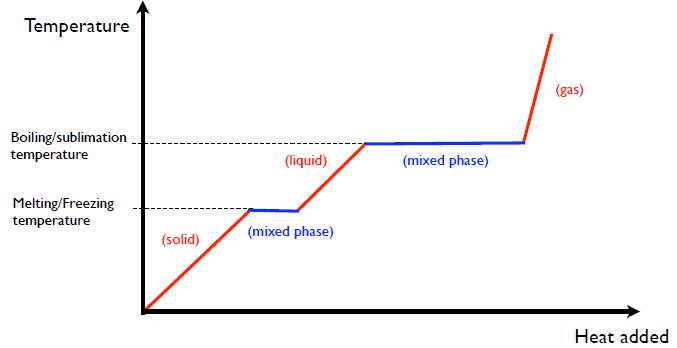
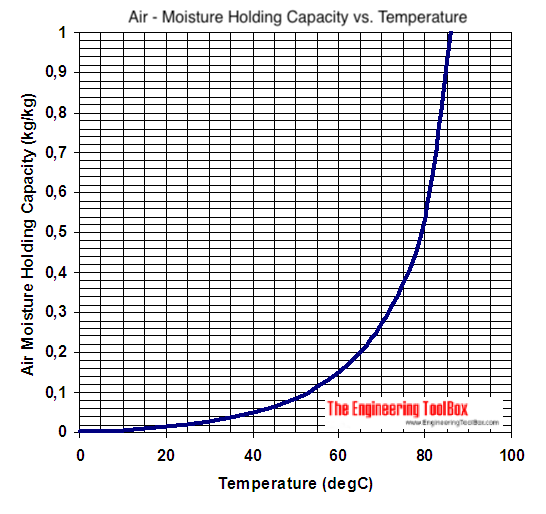
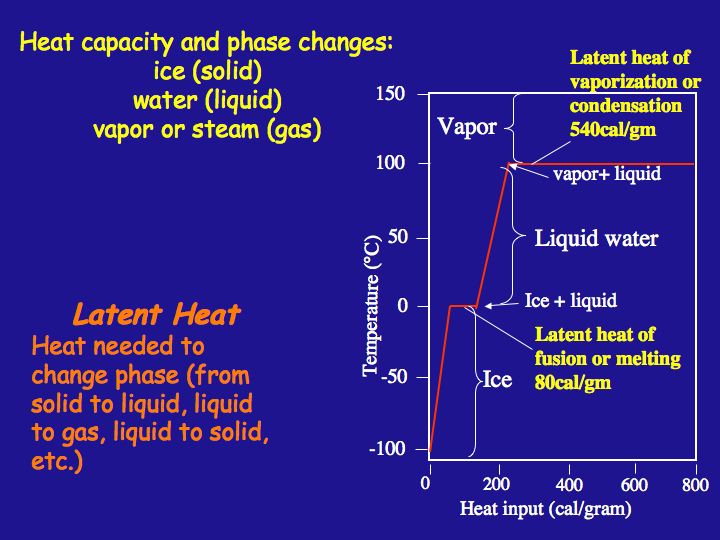
How many J of energy are needed to heat 45.0g of steam from 130oC to 245oC? Why don't you use 4.18 J/goC in this calculation? - Quora
![PDF] Dependence of the isobaric specific heat capacity of water vapor on the pressure and temperature | Semantic Scholar PDF] Dependence of the isobaric specific heat capacity of water vapor on the pressure and temperature | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6c137407717ac2381ee1b2a89743eedede34ab16/3-Figure1-1.png)
PDF] Dependence of the isobaric specific heat capacity of water vapor on the pressure and temperature | Semantic Scholar
![PDF] Dependence of the isobaric specific heat capacity of water vapor on the pressure and temperature | Semantic Scholar PDF] Dependence of the isobaric specific heat capacity of water vapor on the pressure and temperature | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6c137407717ac2381ee1b2a89743eedede34ab16/3-Table1-1.png)