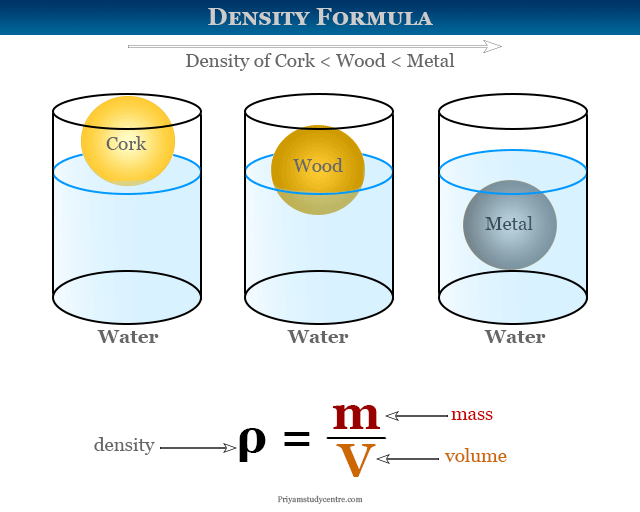
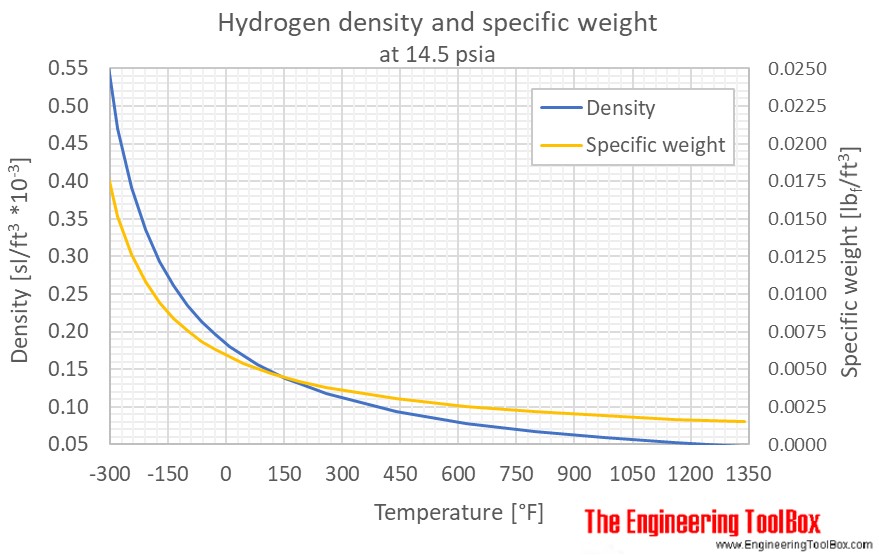
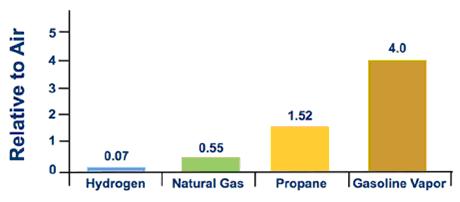
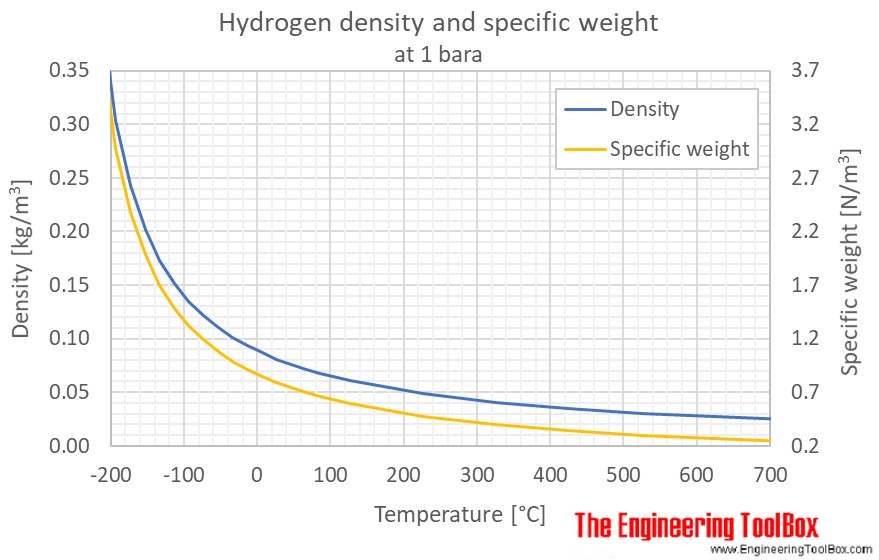
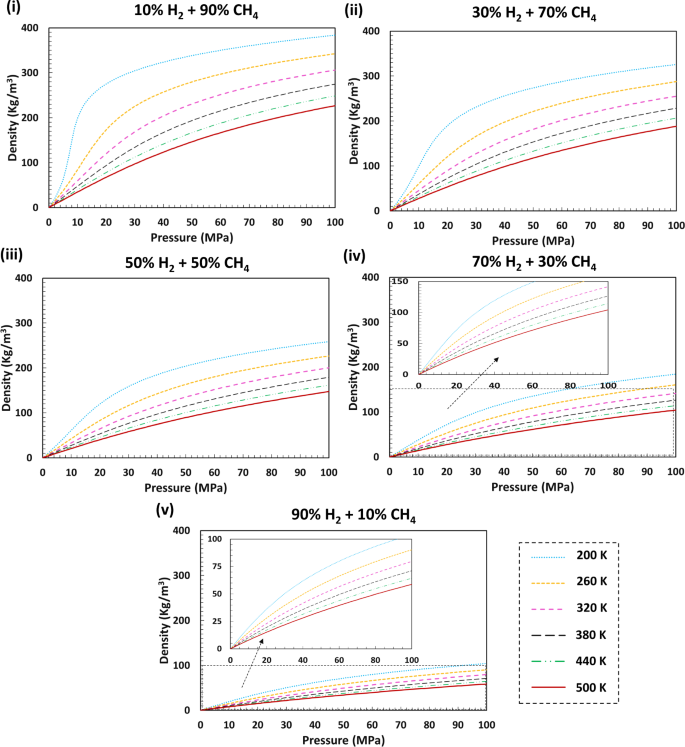
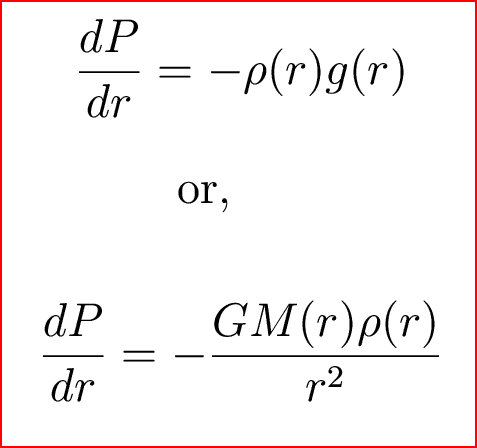Gas Density Formula & Calculation | How to Find Density of a Gas - Video & Lesson Transcript | Study.com
− − − − Pressure versus density of hydrogen gas at ambient temperature... | Download Scientific Diagram

Calculating density and viscosity of a gas mixture in excel · Issue #1695 · CoolProp/CoolProp · GitHub

SOLVED:(a) Calculate the density of sulfur hexafluoride gas at 94.26 kPa and 21 ^∘ C. (b) Calculate the molar mass of a vapor that has a density of 7.135 g / L
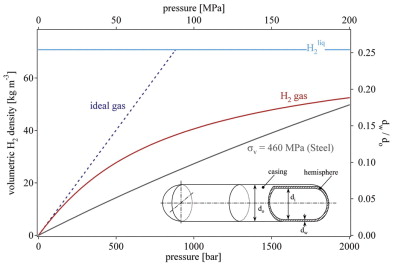
Volumetric density of compressed hydrogen gas as a function of gas... | Download High-Resolution Scientific Diagram


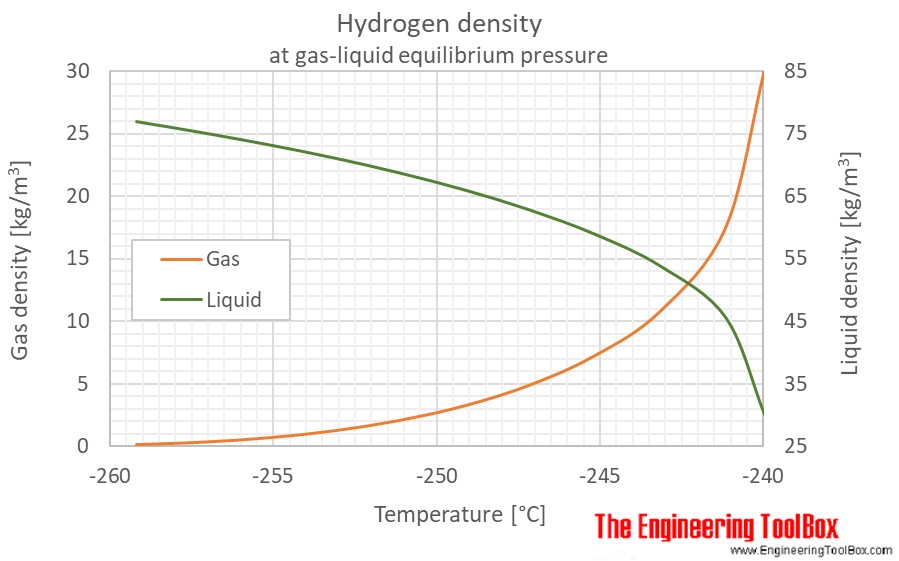

.gif)