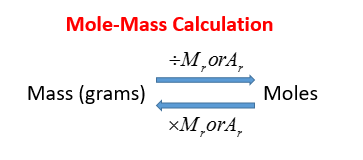Empirical formula and reactant mass Learning Objective: To be able to perform calculation without a problem! - ppt download

Bell Work Date SPI Density Select the best answer. Yes you may use a calculator (1) (2) - ppt download

Determine the empirical formula of an oxide of iron which has 69.9% iron and 30.1% dioxygen by mass. - YouTube

How To Calculate Relative Atomic Mass - What is Relative Mass? How to Calculate Relative Atomic Mass along with FAQs

Calculating reacting masses What mass of iron could be produced from 80g of iron oxide? Fe 2 O 3 + 3CO 2Fe + 3CO 2 (56x2)+(16x3) 2x ppt download