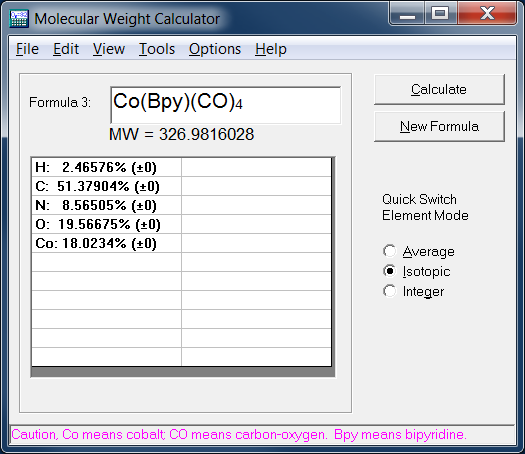
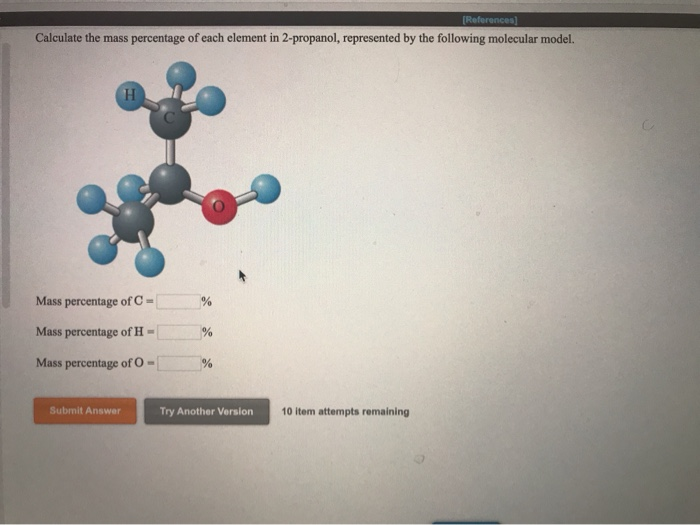
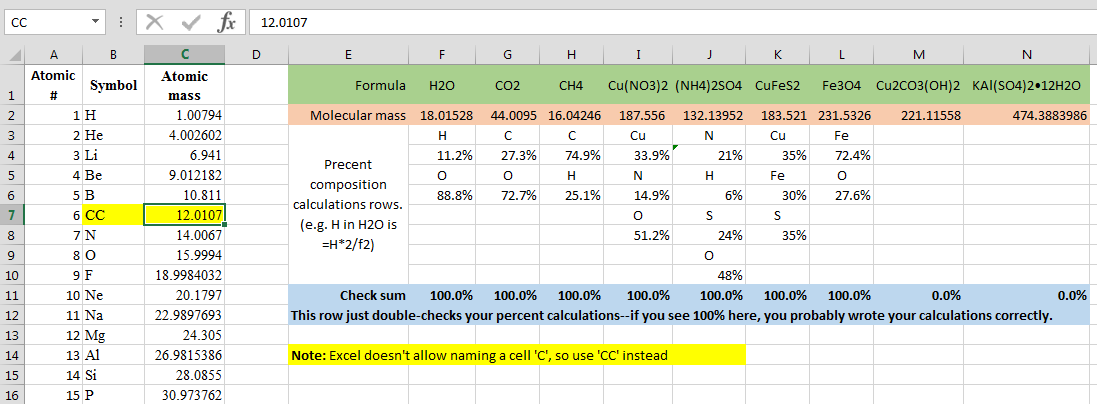
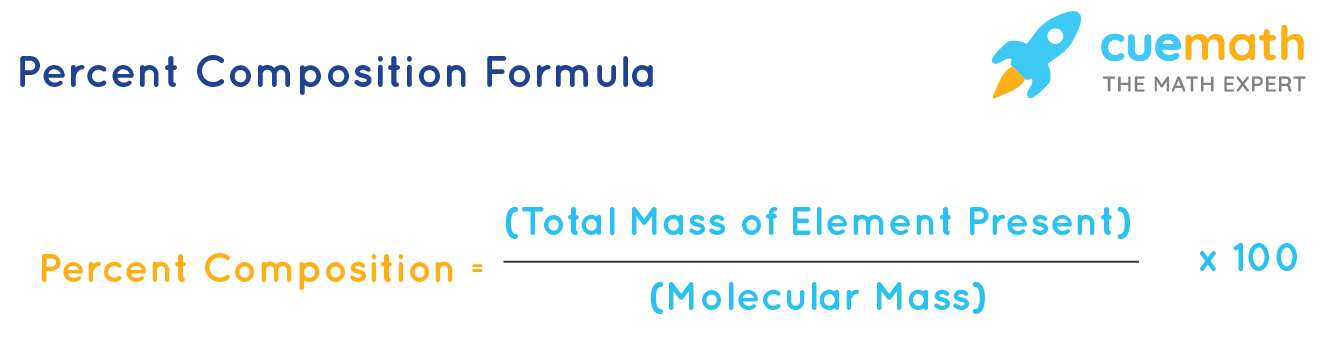The empirical formula of the compound is CHO2 . The molecular weight of the compound is 90. Calculate the molecular formula of the compound.

4.6 MOLECULAR FORMULAS. 1. Determine the percent composition of all elements. 2. Convert this information into an empirical formula 3. Find the true number. - ppt download

Calculating Percent Composition and Determining Empirical Formulas - Video & Lesson Transcript | Study.com
![Calculate the molecular mass of haemoglobin C(3021)H(4780)O(896)N(760)S(12)e(4) and find the mass percentage of iron (Fe) in this molecule. [R.A.M.: H = 1, C = 12, N = 14, O = 16, S=32, Fe=56] Calculate the molecular mass of haemoglobin C(3021)H(4780)O(896)N(760)S(12)e(4) and find the mass percentage of iron (Fe) in this molecule. [R.A.M.: H = 1, C = 12, N = 14, O = 16, S=32, Fe=56]](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/644043869_web.png)
Calculate the molecular mass of haemoglobin C(3021)H(4780)O(896)N(760)S(12)e(4) and find the mass percentage of iron (Fe) in this molecule. [R.A.M.: H = 1, C = 12, N = 14, O = 16, S=32, Fe=56]

How To Calculate Relative Atomic Mass - What is Relative Mass? How to Calculate Relative Atomic Mass along with FAQs



:max_bytes(150000):strip_icc()/mass-percent-composition-example-609567_V2-01-89c18a9d30ea43b494d09b81f7ffefc1.png)