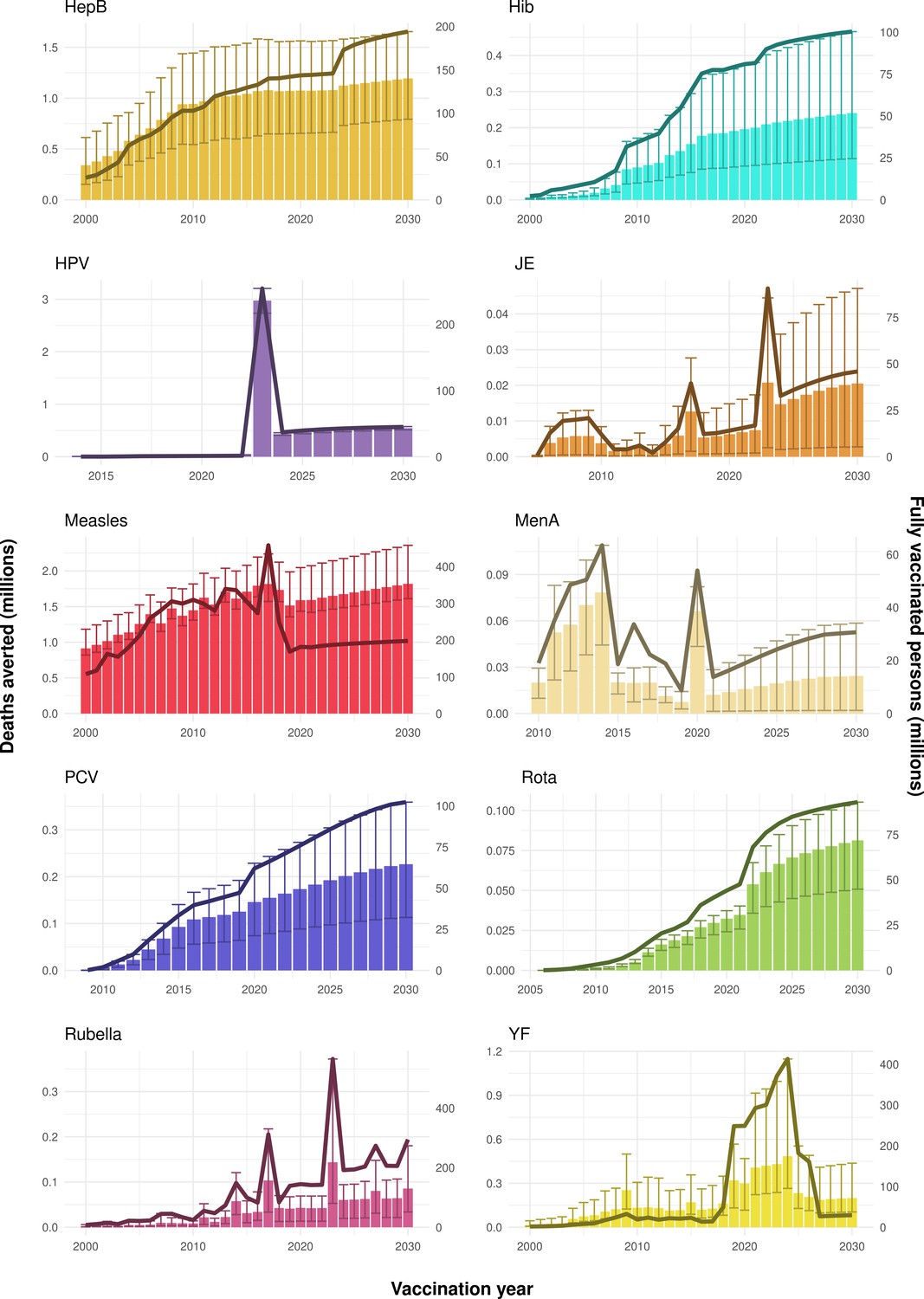
Adenovirus-based vaccines—a platform for pandemic preparedness against emerging viral pathogens: Molecular Therapy

Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial - The Lancet

The paradigm shift in treatment from Covid-19 to oncology with mRNA vaccines - Cancer Treatment Reviews
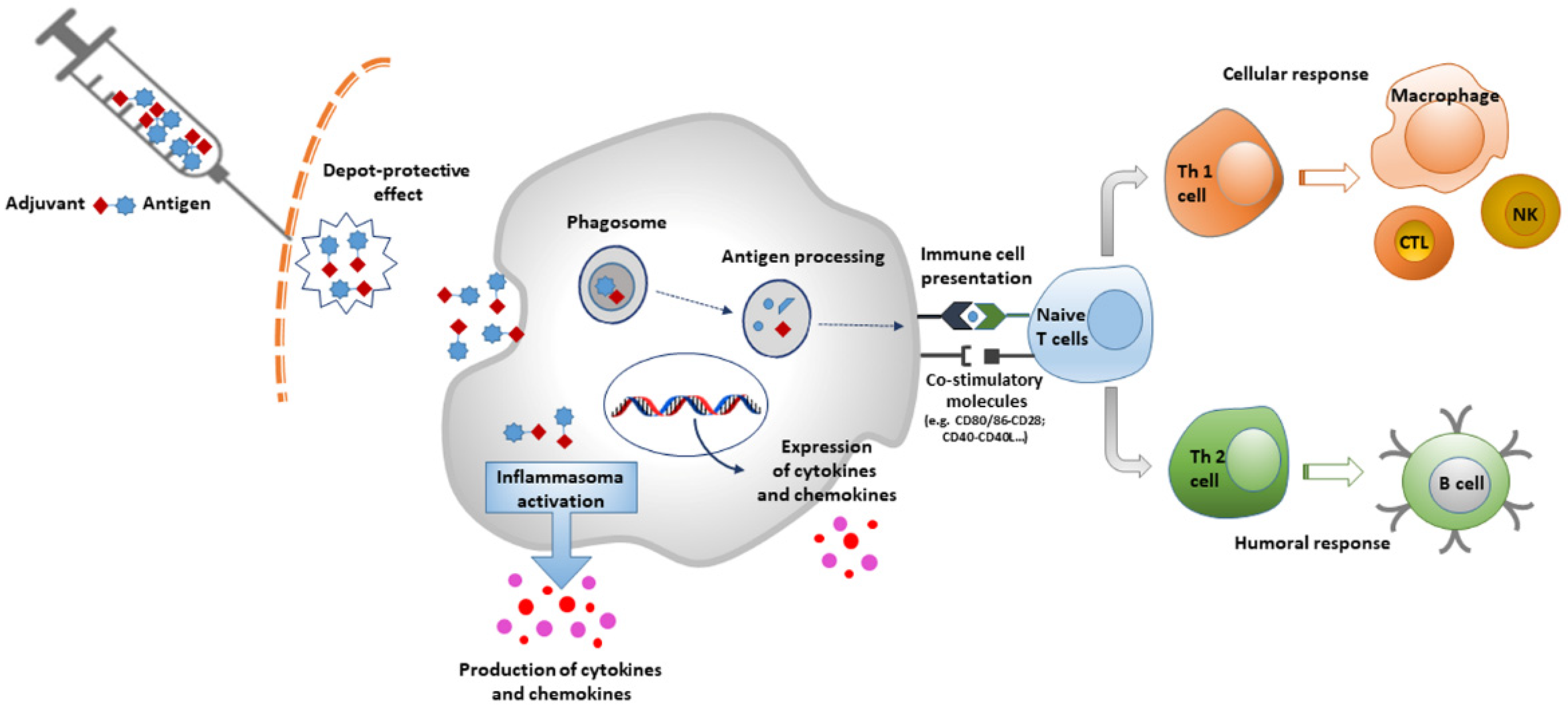
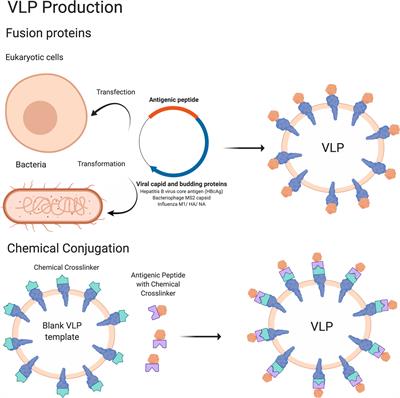
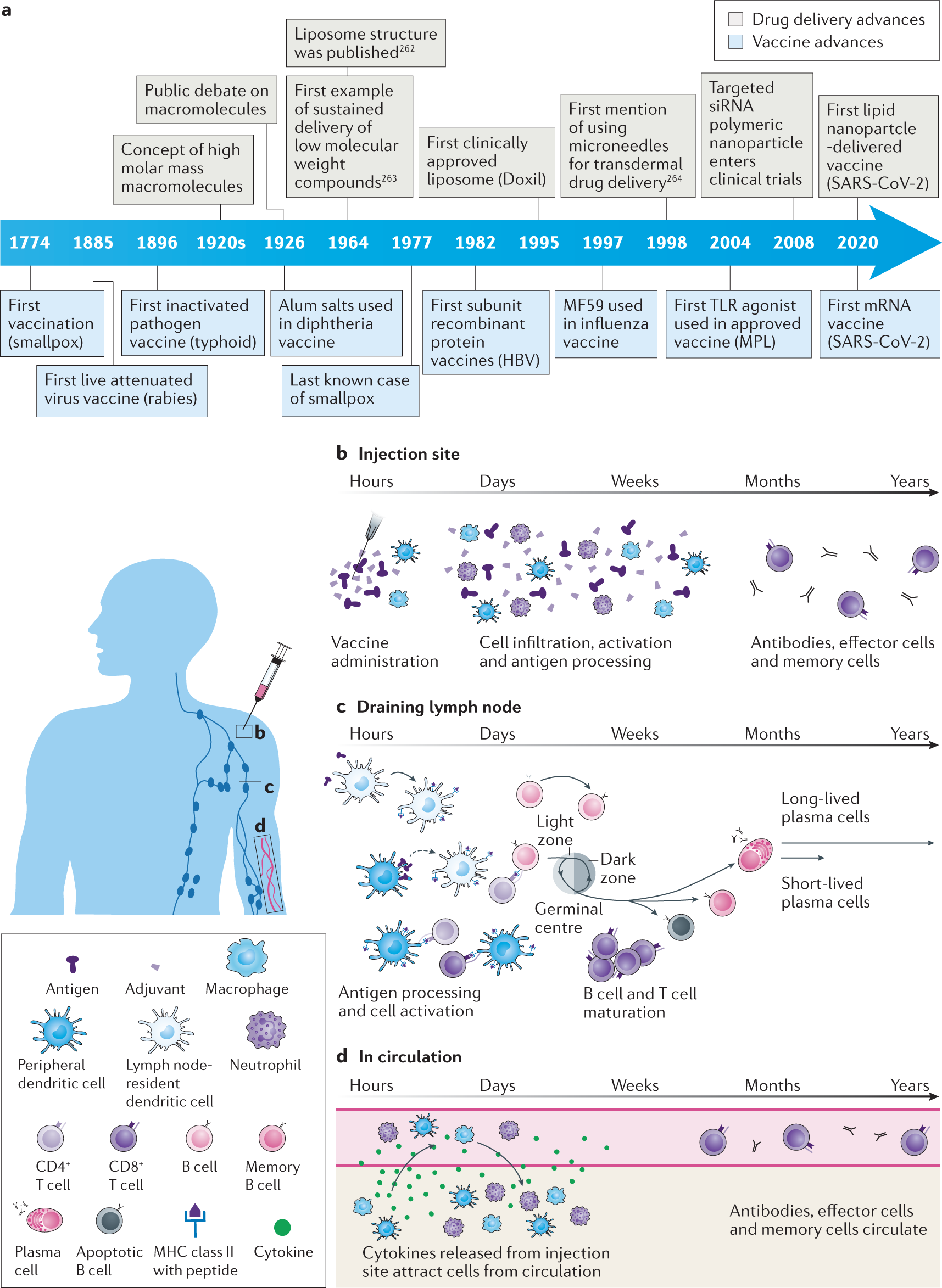
Vaccines | Free Full-Text | An Overview of Vaccine Adjuvants: Current Evidence and Future Perspectives | HTML

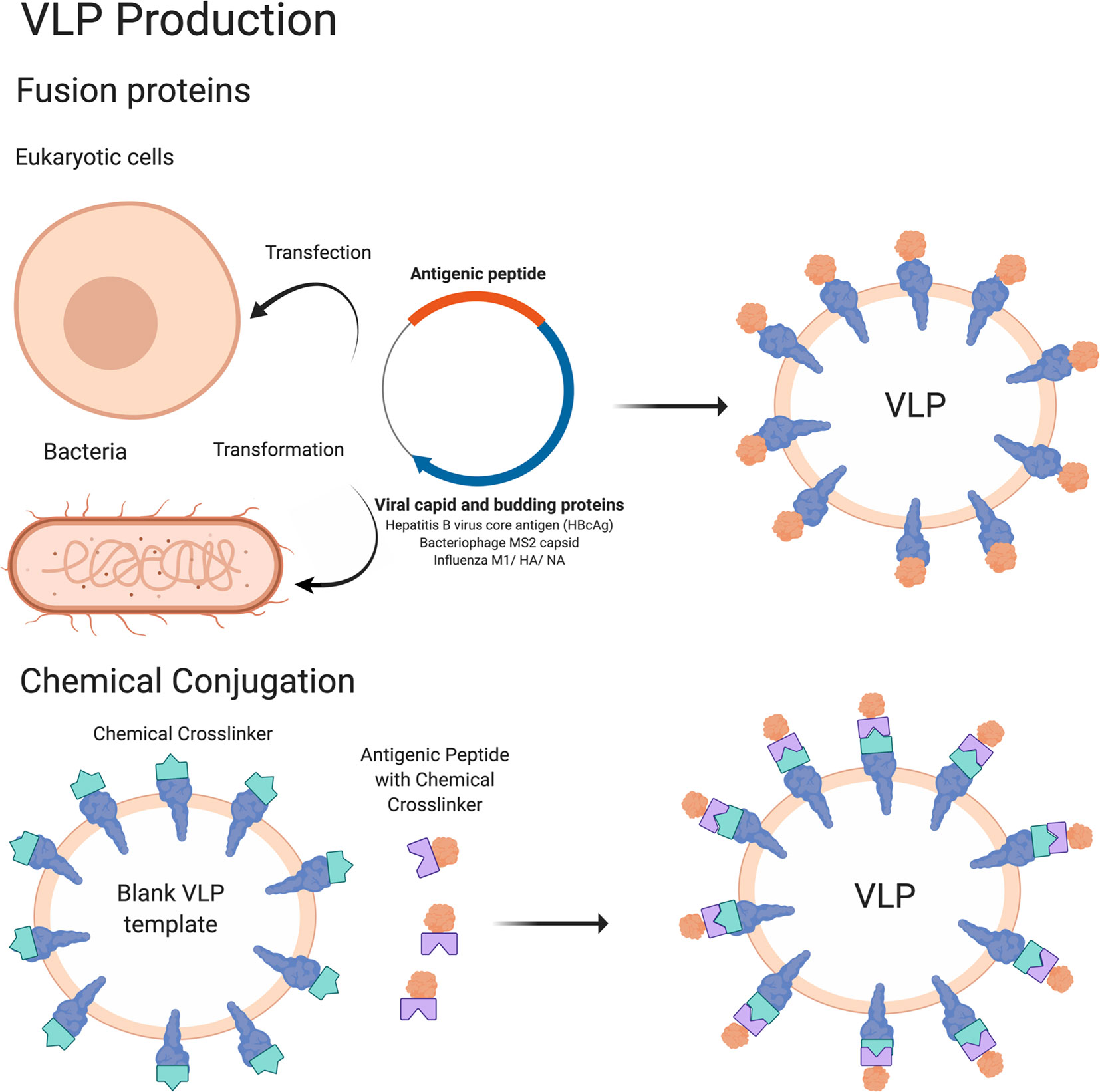
Synthetic Glycans to Improve Current Glycoconjugate Vaccines and Fight Antimicrobial Resistance | Chemical Reviews
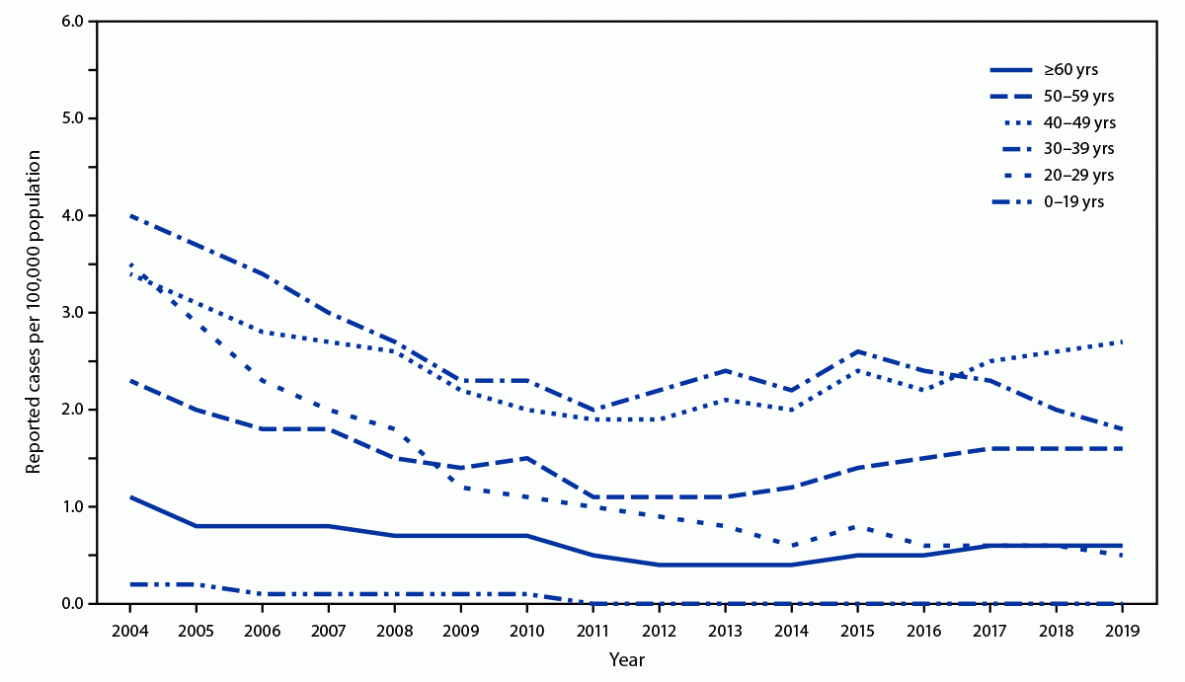
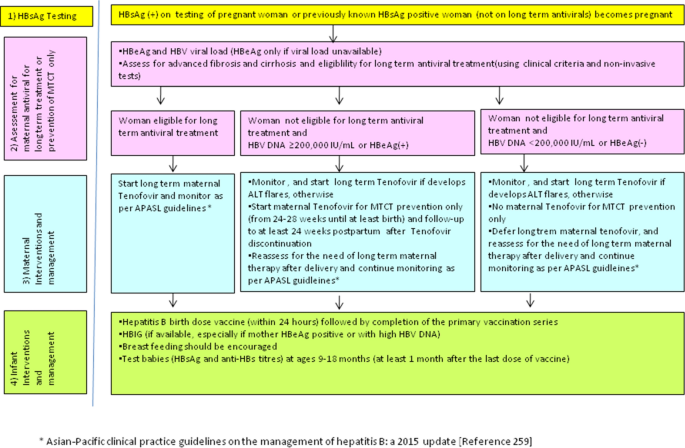
Universal Hepatitis B Vaccination in Adults Aged 19–59 Years: Updated Recommendations of the Advisory Committee on Immunization Practices — United States, 2022 | MMWR
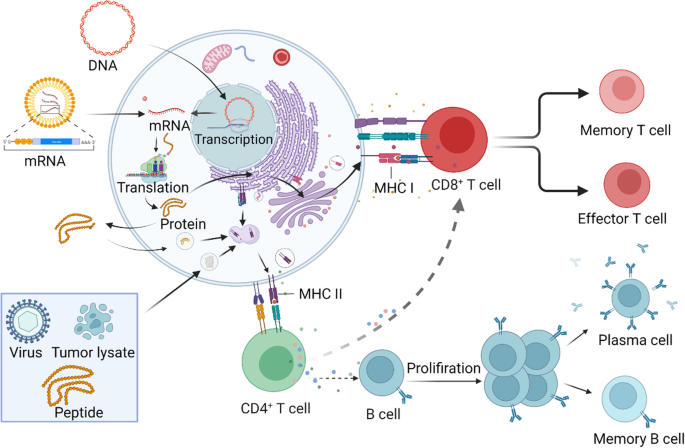
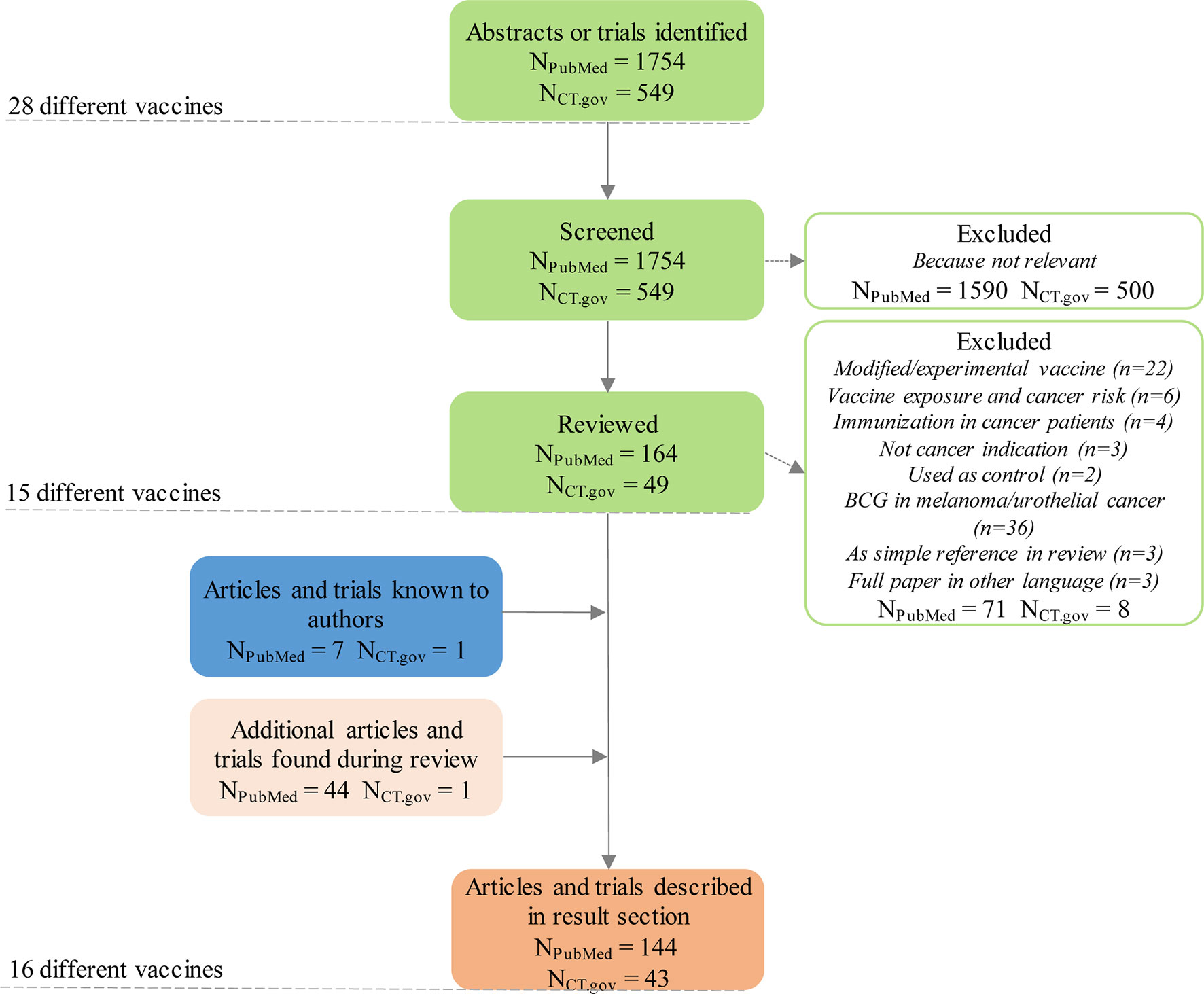
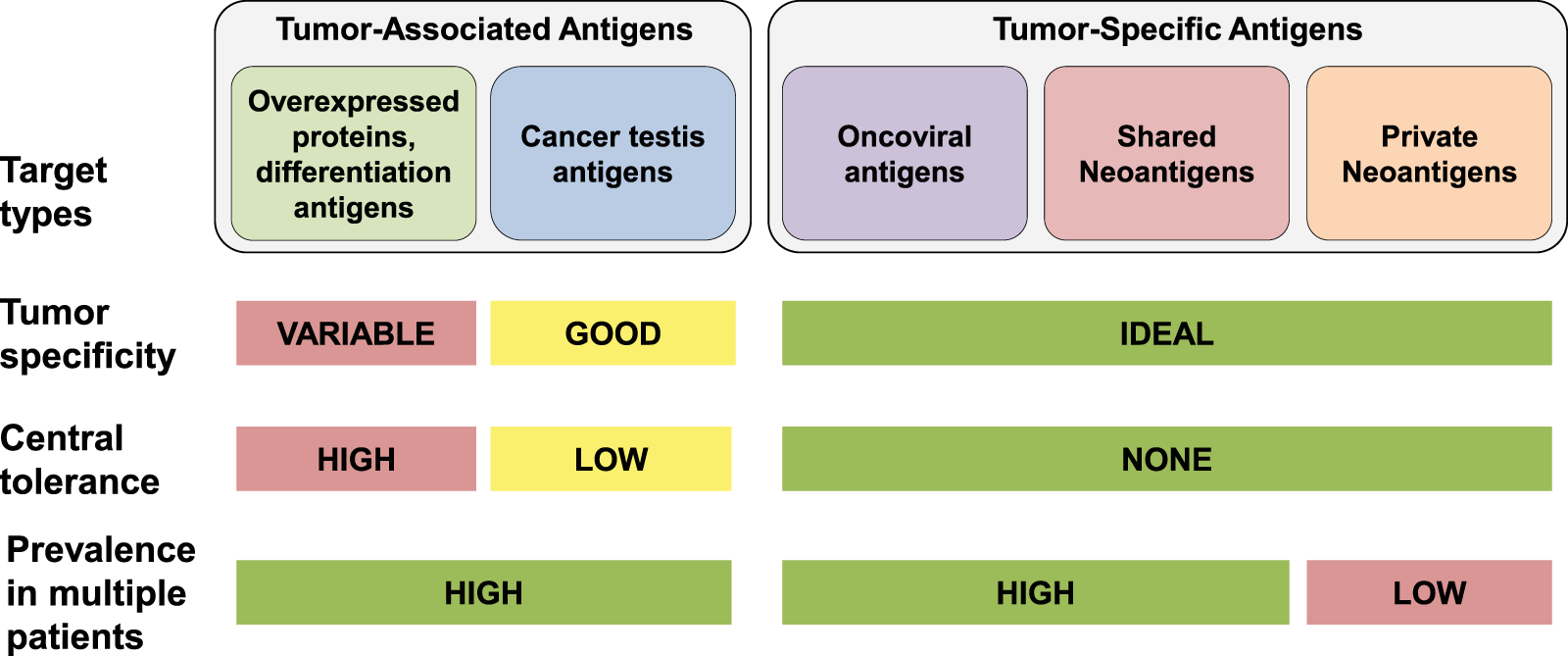
Cancer vaccines as promising immuno-therapeutics: platforms and current progress | Journal of Hematology & Oncology | Full Text